Back رابطة ثنائية Arabic Двойна връзка Bulgarian Dvostruka veza BS Doble enllaç Catalan Dvojná vazba (chemie) Czech Doppelbindung German Διπλός δεσμός Greek Duobla ligo Esperanto Enlace doble Spanish Kaksikside Estonian
In chemistry, a double bond is a covalent bond between two atoms involving four bonding electrons as opposed to two in a single bond. Double bonds occur most commonly between two carbon atoms, for example in alkenes. Many double bonds exist between two different elements: for example, in a carbonyl group between a carbon atom and an oxygen atom. Other common double bonds are found in azo compounds (N=N), imines (C=N), and sulfoxides (S=O). In a skeletal formula, a double bond is drawn as two parallel lines (=) between the two connected atoms; typographically, the equals sign is used for this.[1][2] Double bonds were introduced in chemical notation by Russian chemist Alexander Butlerov.[citation needed]
Double bonds involving carbon are stronger and shorter than single bonds. The bond order is two. Double bonds are also electron-rich, which makes them potentially more reactive in the presence of a strong electron acceptor (as in addition reactions of the halogens).
- Chemical compounds with double bonds
-
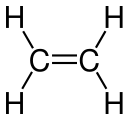
Ethylene Carbon-carbon double bond
-
Acetone Carbon-oxygen double bond
-
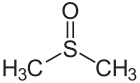
Dimethyl sulfoxide Sulfur-oxygen double bond
-
Diazene Nitrogen-nitrogen double bond
- ^ March, Jerry, 1929-1997. (1985). Advanced organic chemistry : reactions, mechanisms, and structure (3rd ed.). New York: Wiley. ISBN 0-471-88841-9. OCLC 10998226. Archived from the original on 2019-12-10. Retrieved 2020-12-12.
{{cite book}}: CS1 maint: multiple names: authors list (link) CS1 maint: numeric names: authors list (link) - ^ McMurry, John. Organic chemistry (Ninth ed.). Boston, MA, USA. ISBN 978-1-305-08048-5. OCLC 907259297. Archived from the original on 2024-04-04. Retrieved 2020-12-12.
© MMXXIII Rich X Search. We shall prevail. All rights reserved. Rich X Search